Product Development Solutions Comprehensive Solutions for Assay & Kit Development
From concept to commercialization, our team delivers customized solutions that drive innovation and accelerate time to market.
Script Molecular and our team of experts can help medical device manufacturers establish and maintain procedures to control device design and ensure compliance with Design Control requirements as per 21 CFR 820.30.
Key Elements of Design Controls
Design Controls Regulation 21 CFR 820.30/ISO 13485
1. Design and Development Planning:
Establish strategy and plan to define responsibilities for design and development activities.
2. Design Input:
Ensure Design Inputs meet customer requirements and address the intended use. Design Inputs need to be documented, reviewed, and approved.
3. Design Output:
Ensure Design Outputs are clearly defined and conform to
Design Inputs. Design Outputs need to be documented, reviewed, and approved.
4. Design Review:
Formal, documented Design Reviews need to be conducted at
key stages by cross-functional teams and recorded in the Design History File (DHF).
5. Design Verification:
Studies and procedures to verify device design and to confirm that Design Output meets Design Input requirements. Study designs and results of design verification are formally documented, reviewed and approved.
6. Design Validation:
Ensure that device design conforms to defined user needs and intended use through testing under real or simulated conditions. Includes risk analysis and software validation (if applicable).
7. Design Transfer:
Ensure accurate translation of device design into production specifications. Require formally documented, reviewed and approved procedures.
8. Design Changes:
Establish procedures for identification, documentation, validation and/or verification, review, and approval of design changes before implementation.
9. Design History File (DHF):
Establish and maintain a DHF that contains records necessary to demonstrate that the design was developed in accordance with the
approved design plan.
Our 4 Pillars of Product Development
- Assay & Kit Control Development: Design and optimize robust controls for enhanced assay performance and reliability
- Controls & Reagent Kit Development: Develop high-quality control kits and reagents tailored for diagnostic applications
- QC Methods Development: Establish rigorous quality control methodologies to ensure consistency and compliance throughout product life cycle
- Seamless Transfer to Manufacturing: Smooth and efficient transition from R&D to full-scale production, ensuring scalability, quality and regulatory adherence
- IRB Liaison & Clinical Trial Documentation: Supporting Institutional Review Board (IRB) interactions and managing clinical trial documentation to streamline regulatory approval
Product Development Life Cycle
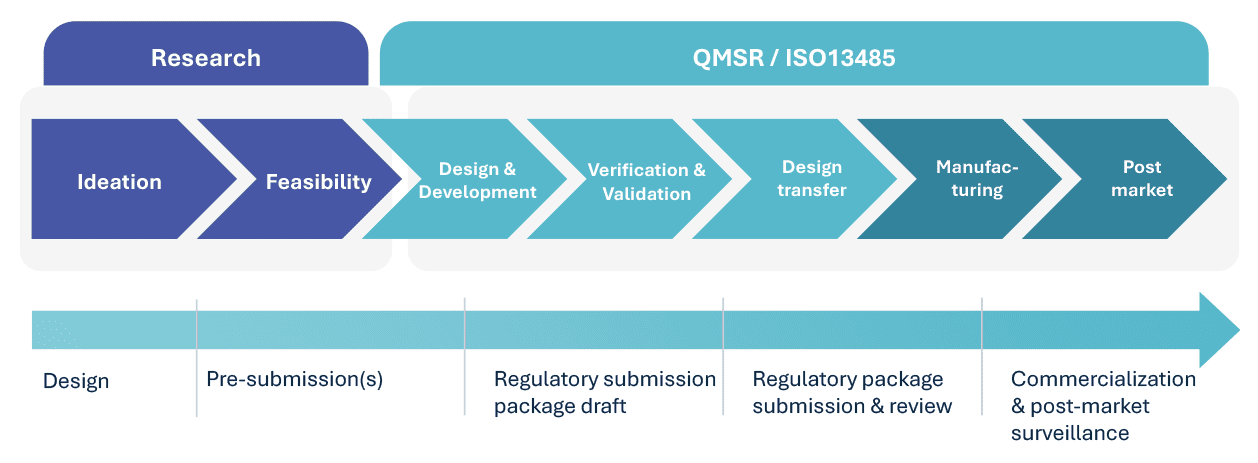
10 steps for Transfer of Design to Manufacturing
1. Design Transfer Plan
21 CFR 820.30
Risk Assessment per ISO 14971
Timelines & roles
2. Final Design Output
Reagent formulation & Kitting
Software for automated analysis
Packaging, Labeling, IFU
3. Develop DMR
21 CFR 820.181
Manufacturing processes
Testing protocols & Specifications
4. Validation
Design Val & Clinical Performance
Pilot Lot production
Usability Testing
Process Validation 21 CFR 820.75
5. Facility and Equipment Readiness
21 CFR 820
Equipment validation for critical processes
6. Training & Knowledge Transfer
Device, Reagents
QC procedures & Acceptance criteria
MDR, Recalls, Corrections
7. Risk Management Integration
21 CFR 820. 30
ISO 14971
Assay, System & Software Risk
8. Regulatory Submission
510(k) package preparation
Compile Manufacturing & Validation Data
9. Full-sale Manufacturing
21 CFR 820.70
Scale-up
Performance Monitoring
10. Post-market Surveillance
21 CFR 820.198
Complaint Handling
CAPA - Continuous Improvement
6 steps for Commercial Manufacturing and Product Launch
1. Market Analysis & Strategy Development
- Market Research
- Pricing
- Go-to-market strategy
2. Regulatory Compliance and Approvals
- Regulatory clearance
- Adherence to GMP
- Compliance with quality standards
3. Manufacturing and Supply-Chain Readiness
- Scale-up manufacturing
- Establish reliable supply-chain
4. Marketing and Branding
- Ascertain costing
- Create branding, packaging & promotional material
- Plan and execute marketing campaigns
5. Sales and Distribution Channels
- Develop relationships with distributors, retailers, partners
- Implement logistics plan
- Customer feedback
6. Post-market Surveillance & Support
- Monitor performance, Customer complaints, MDR, Corrections, Recalls
- Compliance with regulatory requirements
